FDA grants clearance for Philips’ Rembra scanning platform
Summary
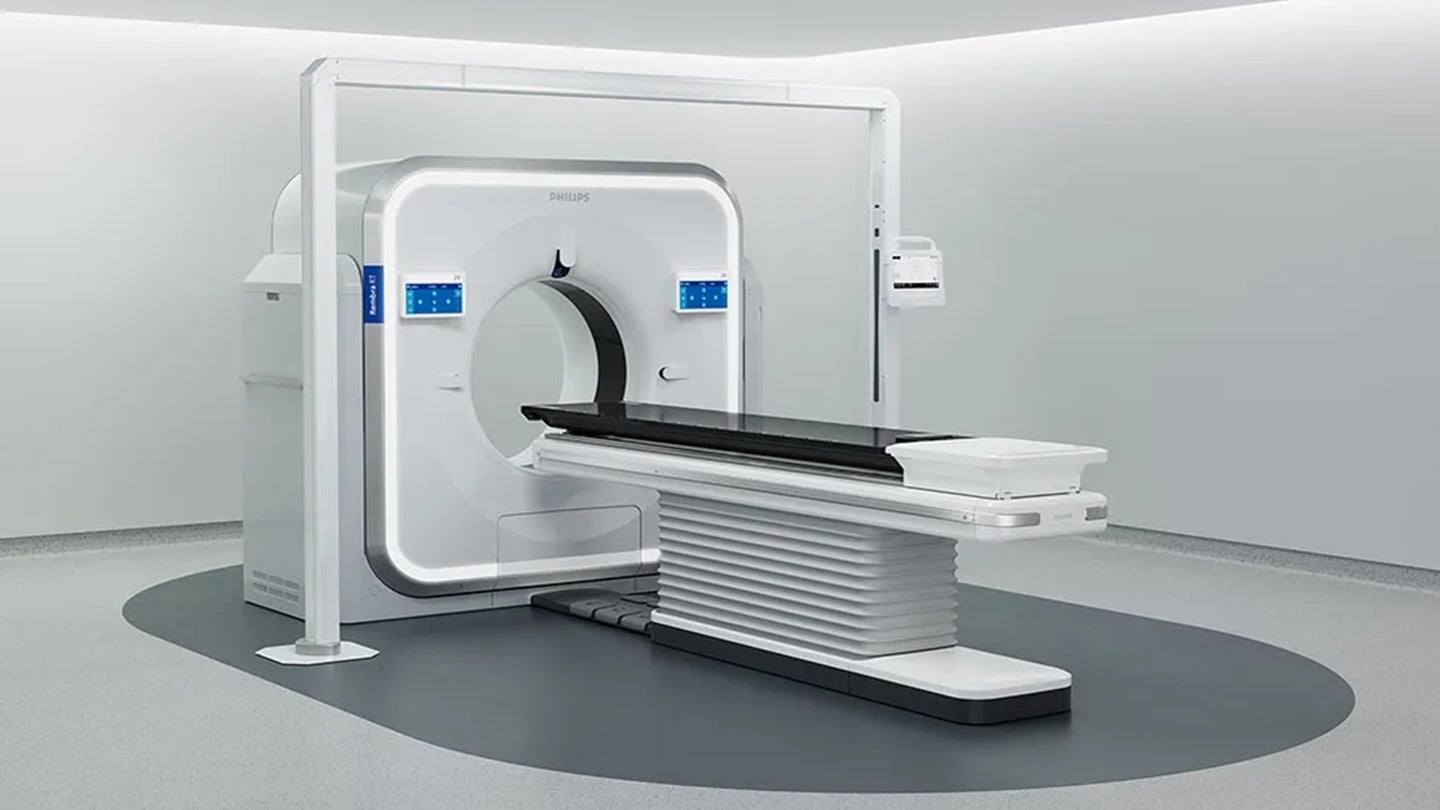
Philips has received the US FDA 510(k) clearance for its Rembra platform of scanning systems, which covers the Rembra CT, Rembra RT, and Areta RT systems.
Description
Philips has received the US FDA 510(k) clearance for its Rembra platform of scanning systems, which covers the Rembra CT, Rembra RT, and Areta RT systems.
Original reporting
AFBytes is a read-only aggregator. Use the original source for full context and complete reporting.
Open original source