Abbott secures FDA approval for Ultreon 3.0 imaging platform
Summary
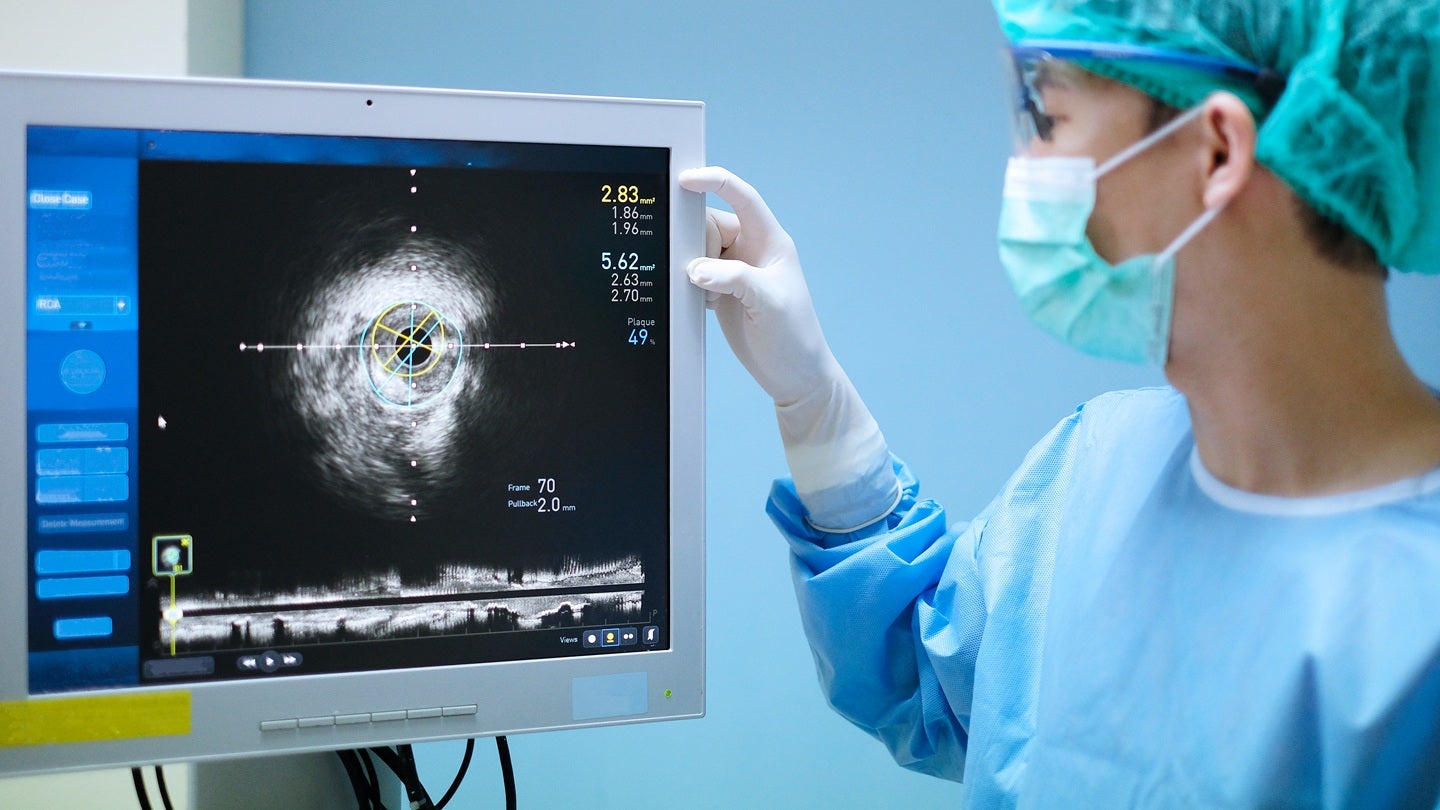
Abbott has received the US FDA approval and CE Mark for its Ultreon 3.0 artificial intelligence (AI)-powered coronary imaging platform.
Description
Abbott has received the US FDA approval and CE Mark for its Ultreon 3.0 artificial intelligence (AI)-powered coronary imaging platform.
Original reporting
AFBytes is a read-only aggregator. Use the original source for full context and complete reporting.
Open original source